
Written by Brad Buecker | Power Plants
Steam is utilized for process heating, mechanical work, and to generate electricity at many thousands of industrial facilities and power plants. Steam purity, quality, and other parameters are widely variable depending upon the application. This article identifies steam terminology and provides several steam-use examples to give the reader a good grasp of general steam requirements for a variety of applications.
Definition – Steam Purity vs. Steam Quality
Confusion often arises regarding the difference between steam purity and steam quality. Purity refers to the concentration of contaminants within steam. For example, high-purity steam contains low part-per-billion levels of impurities such as sodium, chloride, silica, etc. Quality describes the percentage of steam in a water/steam mixture. A steam of 80% quality contains 80% steam and 20% moisture.
Steam Types and Applications
The figure below is a general schematic of steam-generating systems that exist in some form at many co-generation plants.

Many industrial boilers are designed to produce only saturated steam, or “sat steam” in the common vernacular. The boiler imparts the required amount of sensible and latent heat to the water to produce 100% quality steam, but with no additional energy added. Other applications, such as turbine operation, require superheated steam. Outlined below are several important parameters for the choice of saturated versus superheated steam.
Saturated Steam
Many heat exchangers rely on condensing steam or direct steam contact for energy transfer. Sat steam is often ideal for these applications, per factors that include:
- These processes recover most of the steam’s latent heat.
- High heat transfer coefficient
- Condensing steam provides rapid, uniform heating.
Superheated Steam
For some applications, and most notably turbine operation, saturated steam is unacceptable. Upon entry to the turbine, saturated steam would immediately begin to condense, whereupon the moisture droplets in the high velocity fluid would induce serious blade damage. In fact, power turbines are typically designed so that condensation does not begin until the last one or two low-pressure blade rows. A common rule-of-thumb is a maximum of 10% moisture in these stages.
The superheater(s) comprises a network of tubes located in the boiler backpass that collects saturated steam via a series of headers. The additional heat transfer raises the steam temperature above saturation. For example, a once-common coal-fired power plant design produced, at full load, saturated steam with a temperature of 662o F and superheated steam at 1,000o F. Thus, the degree of superheat is 338o.
The major disadvantage of steam production for conventional power units is that most of the steam’s latent heat is lost in the condenser. The maximum net efficiency of older, coal-fired units is (for those still in operation) perhaps no greater than 30-35%, while even modern ultra-supercritical units have net efficiencies of only 45% or perhaps a bit higher. The quest for higher efficiencies (and lower carbon dioxide emissions) has been a key driving factor in the evolution of co-generation and combined cycle technologies.
Large co-gen and industrial plants often have several boilers situated around the facility. Some of these may be waste heat units with unusual configurations. A rather well-known example from the refinery and petrochemical industries are Transfer Line Exchangers (TLE). The heat source is effluent gas from thermal cracking units or syngas production vessels. The TLE cools the gas to prevent further hydrocarbon cracking, while at the same time generating steam for other unit operations. The sometimes unusual configuration of waste heat boilers can lead to potential corrosion and scaling issues that are less common in standard units.
For most industrial steam generators and certainly all steam-based power units, the boiler steam drum is equipped with water/steam separators to minimize entrainment of moisture droplets in the exiting steam.

Numerous factors including separator mechanical damage, drum level surges, and excess impurity accumulation in the boiler water can induce carryover of moisture and contaminants to steam.
Nearly all boilers have some type of makeup water treatment to minimize boiler corrosion and scale formation. The treatment may range from sodium softening for low-pressure industrial units to ultrafiltration®reverse osmosis®ion exchange polishing for high-pressure utility steam generators. Not to be forgotten are impurities that may be present in condensate return. These can include organic compounds; inorganic ions such as hardness, sodium, chloride, sulfate, etc.; and acids and bases. Not only will some of these impurities cause serious boiler damage, but they may also influence steam purity. Figure 2 below is an extract taken from the recent revision of the American Society of Mechanical Engineers (ASME) industrial boiler water guidelines.2
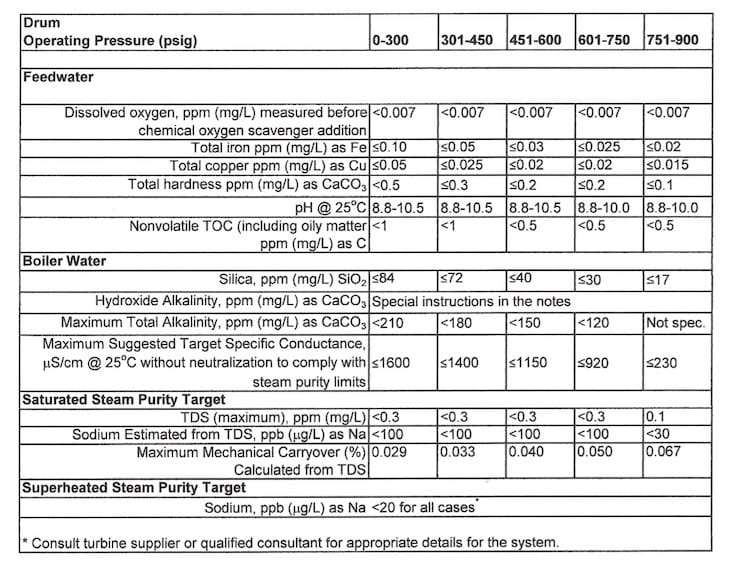
Figure 3. Data extracted from Table 1, Reference 2 – “Suggested Water Chemistry Targets Industrial Water Tube with Superheater”
In large measure, these guidelines were developed to protect steam purity. Moisture carryover to steam is influenced by pressure. This is reflected in the increasingly stringent specific conductivity values at higher pressures. Conductivity is a surrogate measurement for dissolved solids, and the data represents general values designed to maintain the steam sodium concentration below 20 ppb. (For high-pressure power boilers, this limit is less than 2 ppb.3)
Notice also the low limits for TOC (total organic carbon). This issue is particularly important for refineries, petrochemical plants, and similar facilities. In a well-known case, excessive organic leakage into the condensate return for four, 550-psig package boilers induced foam that carried over to the superheaters, which in turn caused overheating and failures on a regular basis.4
Common chemical treatment programs to minimize condensate and feedwater corrosion include ammonia or neutralizing amine treatment to control pH and oxygen scavenger/reducing agent feed to mitigate oxygen attack. As is discussed next, in some applications such as food and beverage production, pharmaceuticals, etc., these chemicals may not be allowed, which requires alternative steam generation or boiler water treatment methods.
Pure Steam
A number of applications require pure steam that has no chemical additives or only compounds that meet requirements of the Food and Drug Administration (FDA) and U.S. Department of Agriculture. Industries that require such steam include:
- Food and dairy. Steam injected directly into food products is known as culinary steam.
- Healthcare, including steam for autoclaves, sterilizers, and perhaps air humidification of clean rooms
- Pharmaceuticals
In cases where conventional boiler steam is permitted, a non-toxic feedwater oxygen scavenger such as sodium erythorbate may be necessary. Other applications might require a specialty boiler to produce high-purity steam free of any potentially hazardous compounds. An electric boiler may be an option, where only a small steam volume is needed that can be produced under carefully controlled conditions. Evolving are electric boilers for larger industrial applications where the energy source is renewable electricity.
Conclusion
Steam production for process heating, mechanical work, and electrical generation will continue to be an essential unit operation of modern process facilities. Numerous steam production methods are available, as are requirements for steam conditions and purity. Boilers and other steam system components require careful design to meet project requirements. Also necessary is conscientious operation and maintenance of steam systems, including makeup water and condensate-return treatment equipment. Examples are well known in which plant personnel focused on process chemistry and engineering to the neglect of boilers and other steam system components. A sudden failure then caused partial or complete plant shutdown and in some cases jeopardized employee safety. Important for any program is reliable on-line chemistry monitoring to ensure that treatment programs are performing properly and to detect upset conditions.5 Boiler tube and other failures have sometimes occurred within hours after onset of a contamination event.
References
- Buecker (Tech. Ed.), “Water Essentials Handbook”; 2023. ChemTreat, Inc., Glen Allen, VA. Currently being released in digital format at www.chemtreat.com.
- Consensus on Operating Practices for the Control of Feedwater and Boiler Water Chemistry in Modern Industrial Boilers, The American Society of Mechanical Engineers, New York, NY, 2021. The complete guidelines are available from the ASME at very reasonable cost.
- International Association for the Properties of Water and Steam, Technical Guidance Document: Steam Purity for Turbine Operation (2013).
- Buecker, B., Koom-Dadzie, A., Barbot, E., and F. Murphy, “Makeup Water Treatment and Condensate Return: Major Influences on Chemistry Control in Co-Gen and Industrial Steam Generators”; presented at the 41st Annual Electric Utility Chemistry Workshop, June 6-8, 2023, Champaign, Illinois.
- Buecker, “Monitoring of Water and Steam Chemistry for Steam Generators”; Chemical Engineering, September 2019.
